Imagine you are Dr. Chen. She thought she had solved a major biomedical problem. After months of carefully engineering a protein to calm overactive immune cells. But when it was injected into animals, their immune systems flared up instead of quieting down. The reason wasn’t poor design or unexpected toxicity. The protein had been contaminated by a substance so common in research and drug manufacturing that it has shaped medicine for more than a century: endotoxins.

These molecules have cost drug developers billions of dollars in detection and removal efforts. In fact, the global endotoxin detection industry is valued at $2 billion.[1] More importantly, endotoxins have sown confusion across biomedical research, casting doubt on studies in fields ranging from COVID-19 to HIV.[2]
So what exactly are endotoxins, and why are they so disruptive?
Endotoxin History
In the early 1900s, injectable drugs were becoming widely used. But doctors began noticing a strange pattern. Even when the right drug was given, patients often developed sudden fevers, went into shock, and in some cases, died. The phenomenon was so common it became known as “water fever”[3]
Eventually, the culprit was traced not to the drugs themselves but to remnants of bacterial contamination. Specifically, fragments of the outer walls of Gram-negative bacteria–later named endotoxins. Crucially, these fragments remain even after the bacteria die, making them hard to eliminate.
At first, doctors didn’t know why endotoxins were toxic, but they quickly saw that removing them prevented the fevers. Later, assays derived from horseshoe crab blood were invented to detect them, a system that is still widely used today, although researchers are developing more ethical alternatives. [4]
For small-molecule drugs, these measures largely solved the problem. But when it came to protein-based drugs, such as therapeutic antibodies, endotoxins proved much harder to control.
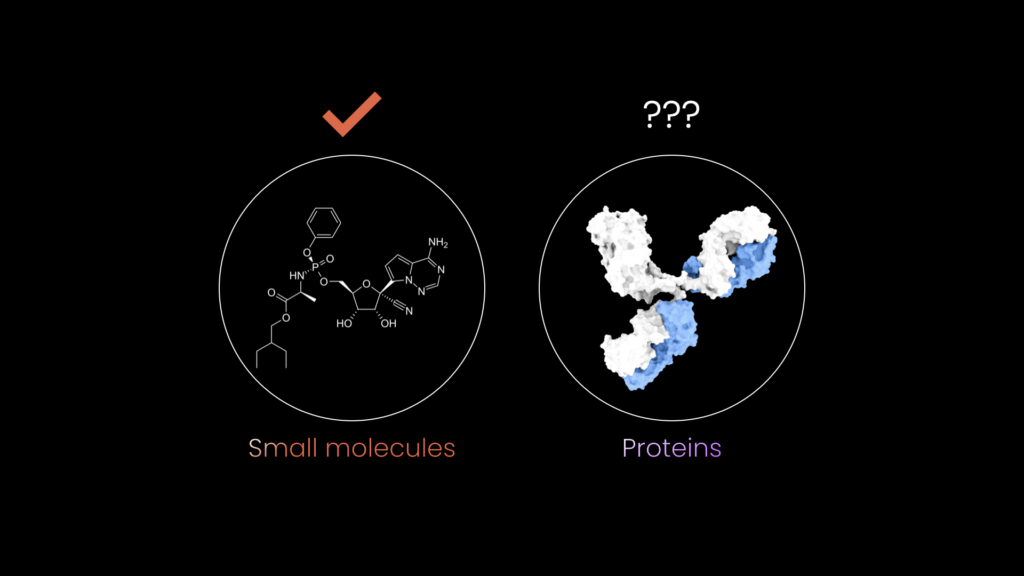
How Endotoxins Trigger the Immune System
A major breakthrough came in 1998, when Bruce A. Beutler discovered that immune cells carry receptors called TLR-4. Endotoxins bind directly to these receptors, setting off an aggressive immune response. In other words, endotoxins aren’t inherently poisonous, the danger comes from how our immune system reacts to them. [5]

Beutler’s work reshaped our understanding of immunity and earned him a share of the 2011 Nobel Prize. But it also opened a new challenge. As researchers investigated TLR-4, they found that proteins with no apparent immune role sometimes triggered it. To this day, debates continue about whether proteins from HIV, dengue, or even COVID-19, can directly activate TLR-4 [2]

So, are these proteins truly dangerous, or simply dirty?
Why Proteins Are Confounded With Endotoxins
The answer lies in how proteins are produced in the lab. The most common system for making recombinant proteins is E. coli, a Gram-negative bacterium. Unfortunately, endotoxins stick stubbornly to proteins. Purification tags or equipment can make the problem worse. They can even disrupt protein folding and function, complicating both research experiments and drug production.
Reusable lab equipment, such as purification columns, compounds the issue by spreading contamination across experiments and entire facilities. What begins as a microscopic impurity can ripple outward, misleading results and stalling progress.
Strategies for Clearing Contamination
So how do scientists fight back? Sure, there are many purification strategies that take advantage of endotoxin properties, such as Polymyxin B.
But every purification step comes at a cost: some of the valuable protein product is lost. For manufacturers, this inefficiency is a serious problem.That’s why prevention is the focus of our strategy. Careful choices, from using low-endotoxin plastics, to pre-treating plasmids, to designing cleanroom protocols, can dramatically reduce contamination risk. We use mammalian cell lines. Though more expensive than E. coli, produce proteins with fewer endotoxin issues and more human-like folding.
Lessons Learned
At Sino Biological, a dedicated production line was designed around these principles, from the choice of materials to clean-in-place protocols. Our new line of products, Propure, achieves endotoxin levels up to 0.01 EU/mg, 100 times lower than industry standards.
But the story of endotoxins is larger than any one company. For over a century, these bacterial remnants have shaped medicine and science. Each chapter of our story didn’t just improve our ability to control endotoxins, it also revealed just how insidious the problem is. The lesson is clear: the deeper we look, the more we see. And only then can we begin to truly solve it.
Visit https://www.sinobiological.com/news/propure-endotoxin-free-proteins for more information
